The Stoplight Protocol

The stoplight protocol is a protocol that I made during my time researching arbuscular mycorrhizal fungi. Its goal is to make the fungal structures that inhabit the cells of the plant visible under the microscope so they can be quantified. This quantification can then be used as data points to describe how well a certain plant is inoculated. The following are several benefits to this protocol that were intentionally designed.
Environmentally Sound: All reagents are able to either be recycled into a new batch or are able to be safely poured down the drain if neutralized properly. All resultant compounds are safe for the environment at quantities relevant to this protocol.
Efficiency: This specific method is more efficient that standard methods and yields clearer results much faster by a factor of around 3x the rate. It is also designed to be done in batches that scale with the size of equipment used.
Universal: Because each sample is unique, this protocol provides a quantitative measurement of completion instead of a qualitative one like time. Timing specimens does not result in uniform results whereas a quantitative approach does.
More Safe: Although many of the reagents used in this protocol are not particularly safe, the protocol as a whole is comparatively safer than standard methods.
Easily Accessible: All reagents are easily accessible and cheap.
There are of course some limitations to this protocol, and they are as follows:
Unsafe Stain: Trypan blue is a carcinogen and should not be poured down the drain. There are unfortunately no safer stains that can be used alternatively.
Quantitative Nature: Because the protocol is quantitative, it is up to the person doing it to accurately interpret their own progress. Interpretation is made to be a simple and standardized as possible.
The following is the unannotated protocol. It is made to be as straightforward as possible, and should be used as a foundational guide. The annotated protocol will be available below and will extrapolate on each step. It will also provide instructions for analysis following preparation.
Unannotated Protocol
*The following must be done in a fume hood
*The following is designed for woody roots of all genera. If you are processing non-sclerotized roots, do not look for a color change, instead incubate for five minutes. Only one KOH treatment followed by an H202 treatment is necessary and any more risk poor visibility and poor integrity.
– Sort samples by species and place each sample in a biopsy cassette and label with permanent marker directly on the cassette.
– Wash the samples through the cassette with water to remove debris.
– Create a 1.75 molar solution of KOH proportional to double the absolute size of the reaction vessel (ex. a 500mL beaker would require 1000mL of solution).
– Create a volumetric solution of 33% glycerol, 33% lactic acid, and 33% water. Add an additional 0.05% of the total volume in grams of Trypan blue. (ex. 100mL of solution would require 5g of trypan blue powder).
– Place labeled samples and KOH solution in reaction vessel. The amount of samples per batch is irrelevant so long as they are all submerged. Cover the vessel with parafilm but allow space for gas to exchange.
– Heat the solution at boiling temperature until it is a deep red (or brown/black). The temperature is not of specific relevance so long as the solution is boiling.
– Immediately place the solution onto a mechanical agitator plate at as rigorous of a speed as possible.
– Continue agitating the solution until the solution is cool enough to handle.
Decant the KOH solution into a collection flask while preserving the samples. Pour in an equal volume of KOH solution and repeat steps 4-6 until the solution is a translucent red.
– Decant the KOH solution into the collection flask and wash the samples with water. The wash water can also be added to the collection flask.
– Submerge the samples in a 5% solution of H2O2. Heat until the solution at boiling temperature until a deep yellow color develops, followed by agitating until cooled. Note that the solution will be foamy and can potentially overflow if unchecked.
– Decant the H2O2 solution down the drain. Almost all of the H2O2 will have decomposed via the heating and reaction, so it will be safe to do so long as the pH is close to neutral. Wash the samples to remove foam.
– Perform an additional KOH treatments until the solution stops developing red and instead develops green/yellow. Do not expose the samples to KOH after this occurs, otherwise they will become too structurally unsound to analyze.
– Wash the samples and submerge them in a 7% solution of HCl. Allow the samples to acidify for five minutes. Decant HCl into collection flask. *Note that this HCl can be used to neutralize the KOH utilizing stoichiometry. The resultant KCl is neutral and there are no other harmful chemicals. Test the pH before disposal and dispose with a running tap.
– Wash the samples and submerge them in the lactoglycerol stain. Heat the stain to boiling and let the samples cool. Check the samples and repeat until acceptably stained.
*Note that the solution may smell like cherries. Do not inhale fumes of the solution as they are mildly carcinogenic after chronic exposure.
*Note that the lactoglycerol solution can be collected and reused. *Note that the samples can be stained at room temperature overnight instead of quickly boiled.
*Analyze samples or otherwise refrigerate them in water until analyzing. They can theoretically be stored indefinitely but may lose color over prolonged storage.
Annotated Notes
Everything that I chose throughout this protocol was for a very specific reason that was backed up by data. I think that I have a very good understanding of the theory behind each step, although my primary goal was to make something effective regardless if I understand why. Here I will describe my reasoning for why each step is the way it is.
Basic Theory
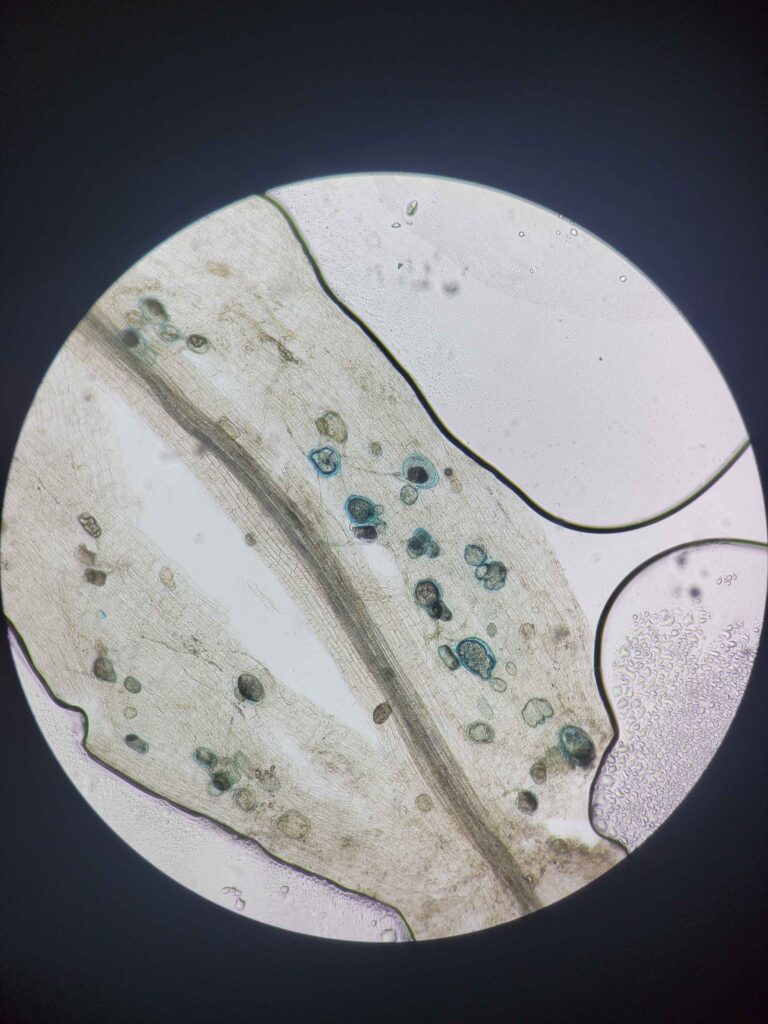
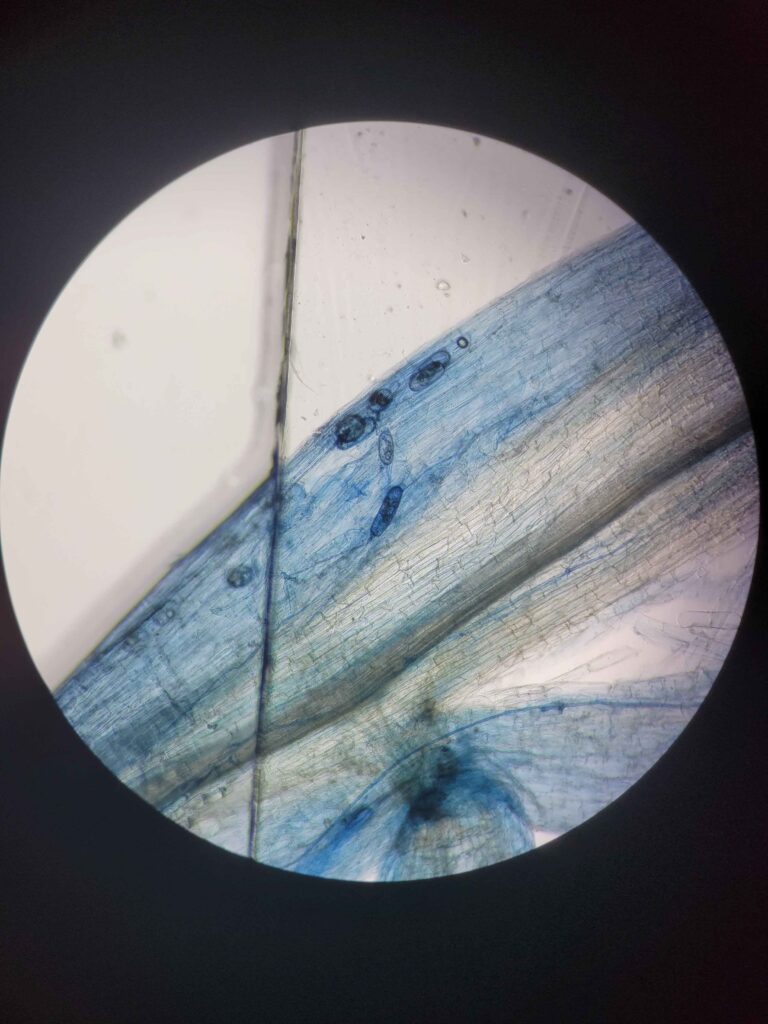
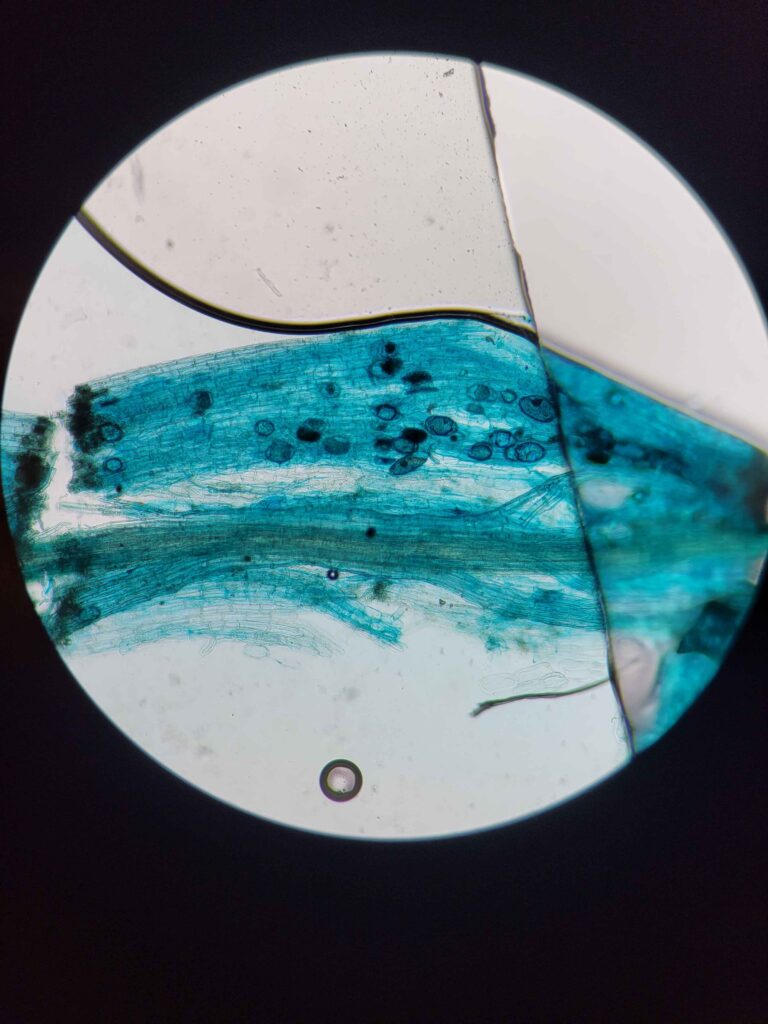
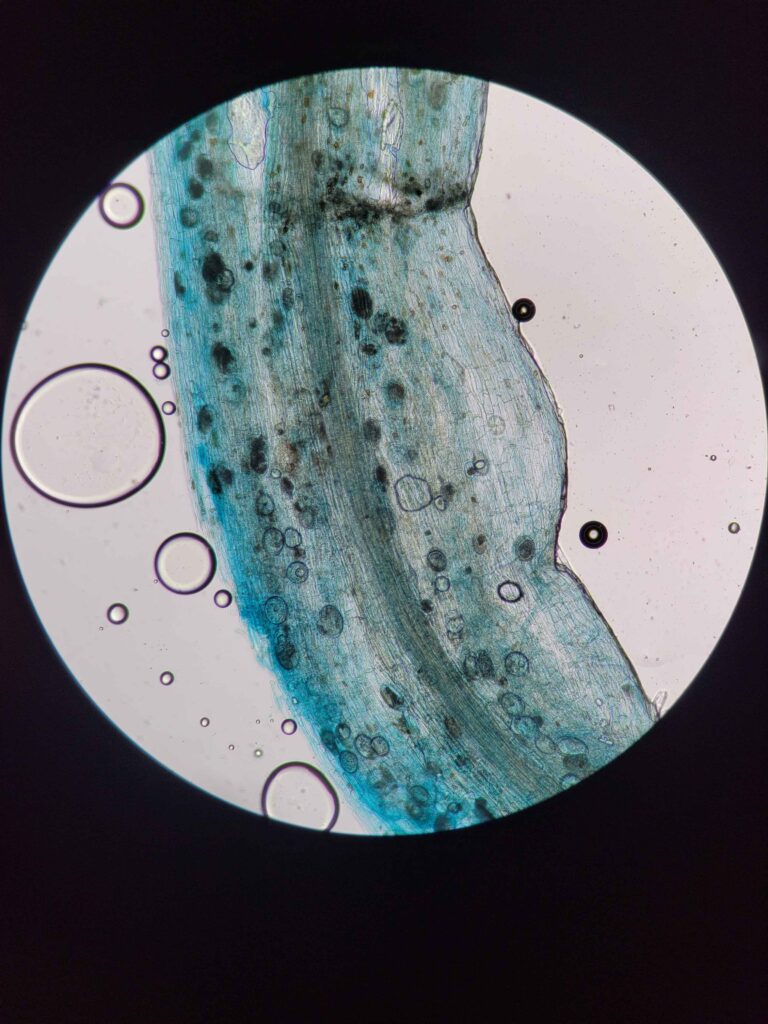
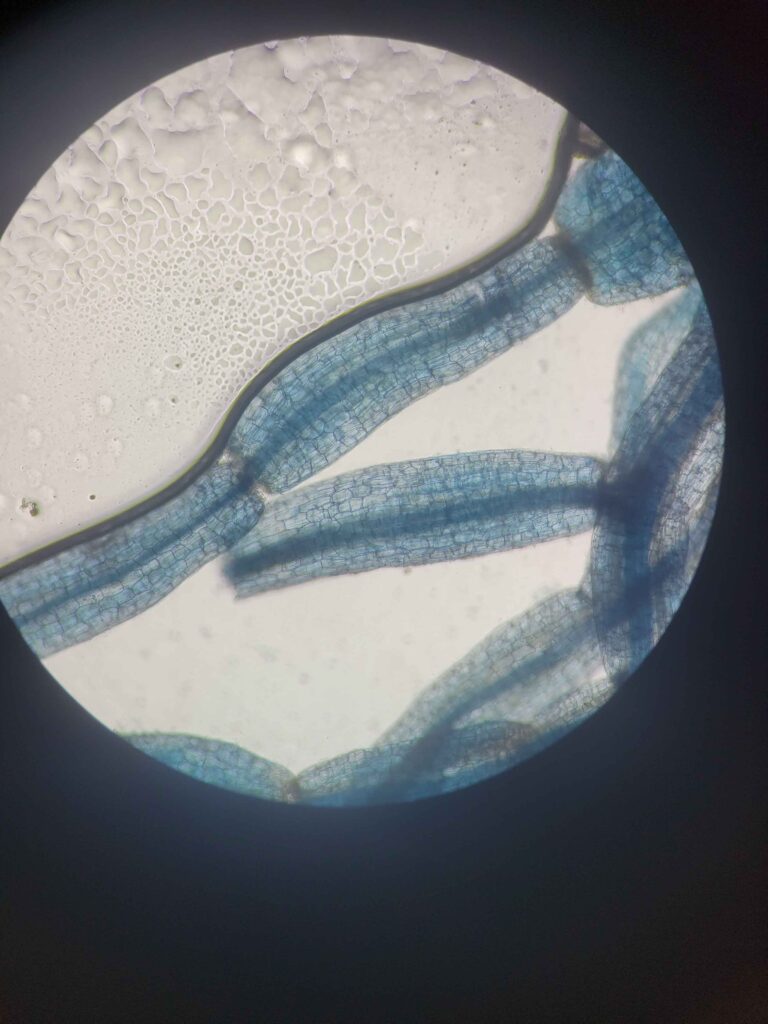
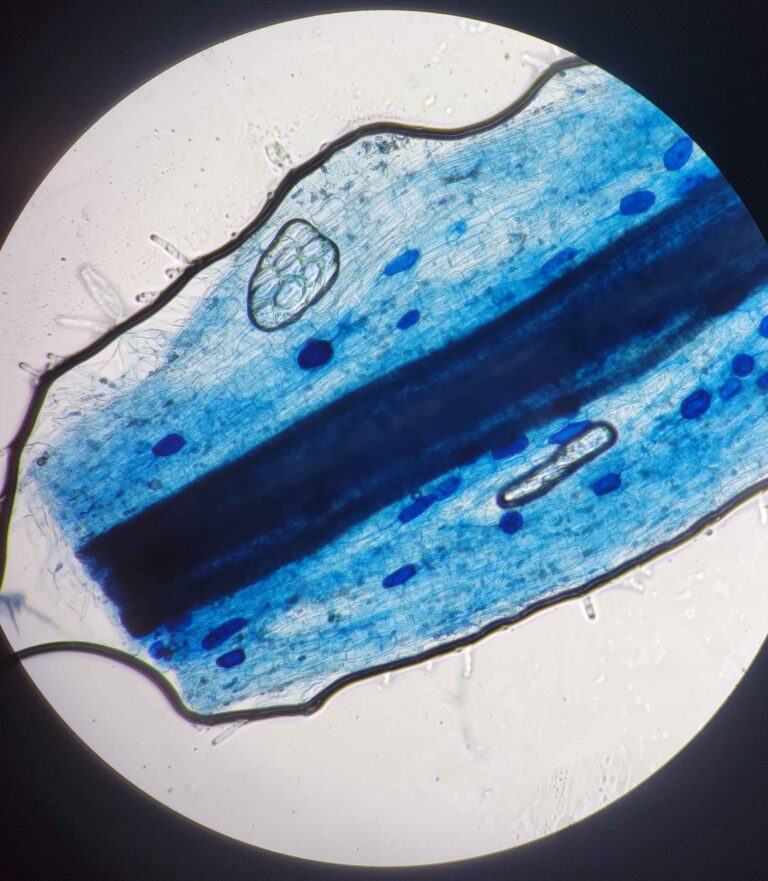
The protocol works via breaking down and evacuating the contents of the plant cells while keeping the fungal cells more or less intact. The fungal cells will be damaged enough for the stain to stick but not completely evacuated. This results in a clear plant tissue scaffold with dark blue splotches of fungal cells.
My theory as to why this works has to do with the fact that fungal cells are surrounded by very strong chitin, which prevents them from being completely evacuated. Plant cells are only protected by cellulose, which is more damaged by strong bases.
Lactoglycerol Trypan Blue Stain
This solution was designed specifically to make the stain stick to the fungal cells and not the cleared plant cells. Trypan blue stains some cellular proteins, and only those that have a damaged membrane (it cannot stain live cells). Hence, the plant cells are mostly entirely evacuated of proteins (most of the remaining proteins will be denatured by the strong base, although some do remain.) while the fungal cells still have enough intact proteins to be stained much darker than the surrounding plant tissue.
Trypan blue is the superior stain in this regard, although direct blue is comparable. Chlorazol black E is also potentially viable but is far worse in terms of contrast. Trypan blue yields the most superior contrast.
The lactoglycerol portion of the stain is necessary as a mounting medium. The stain will not penetrate and stick without it. Solutions of purely aqueous stain result in extremely poor staining and extremely low contrast, especially if not assessed directly after staining.
Usage of Color as a Clearness Indicator
I specifically utilized color as an indicator of clearness because it is universal among all woody species. Each species of plant is cleared at a different rate that is often unpredictable. Utilizing time as an indicator for clearness doesn’t account for these differences and will leave some samples either far too opaque or far too overcleared. The process always ends when the solution turns yellowish green, but the time it takes does not correlate with clearness.
The color differences are due to tannins that are released by the cells as their contents are evacuated. Some species have more or less tannins, and some release them at different rates. The change in color will alway be the same regardless of rate or concentration.
As stated in the protocol, some plants will require more treatments with KOH until the color changes. This is to be expected in larger or thicker samples as well.
Usage of KOH as a Strong Base
A strong base is of course necessary for this protocol. There is no other known way to analyze colonization without expensive and intricate machinery and luminescent dyes. KOH is the best option for this because it is readily available and cheap.
NaOH can be used alternatively with extremely similar results. KOH is marginally better because it is a larger molecule and “attacks” the cells more violently. Note that if NaOH is used that the molarity has to be adjusted to slightly higher to account for this difference. I do not have data as to what extent.
A general rule of thumb for the KOH solution is to just do 10% weight by volume of KOH. I typically make a liter of solution for each batch, so I would use 100g of KOH granules. Quite frankly it does not have to be precise as long as the concentration is in that ballpark. It is not particularly troublesome to make more KOH solution than you need as it can be used in future batches.
Heating and Agitation
Heating the solution is of course beneficial because it speeds up the rate of the reactions between the strong base and cellular components and makes them far more vicious. It is possible for this reaction to be done at room temperature, but it takes several hours.
There isn’t any concern of overheating the samples as the temperature will plateau at boiling. High heats do not degrade the sample, rather, they often come out with higher contrast. There is no risk of “cooking” them, and I suggest holding them to as high of a temperature as is safe and possible.
Placing the samples in an autoclave for about an hour is also effective in terms of quality, although it takes much longer and is less precise. It was far harder to control when heating and agitation was applied and for what lengths. I have limited data on this method because it was impractical to me, but it may be useful for somebody who is doing this as a side task and does not want to constantly monitor a boiling hot plate.
Agitation is a key component of this protocol. Agitating the solution has multiple modes of action, including removal of neutral components, clearing the cells via mechanical removal, and increasing the rate of reactions. This drastically speeds up the protocol in addition to the heating.
Any form of agitating can be used, although I find that a gyrating mechanical agitation table is the most effective. Magnetic stirring was ineffective due to the stir bar getting knocked away by the cassettes, and when it did work, it was not very powerful. A suspended anchor agitator may be more effective, although I did not have that equipment available and have no data on it. Vibrating agitators are also largely ineffective. I have no data on rocking agitators, although I hypothesize that they would be the most superior agitator if the vessel can be used in it without spilling.
Addition of H2O2
The usage of an oxidizer in a protocol like this is quite nonstandard, but I find that it is useful for obtaining clearer samples with a higher contrast between cells. I specify H2O2 because other oxidizers are too violent for the structure of the cells. HClO4 at low concentrations can be used as well, although my data on the exact concentration is limited. It will sometimes result in “mushy” or overcleared samples. The mixing of residual KOH in the samples and HClO4 can also release gaseous chloride compounds, which although is generally not that big of a deal, is not necessary given a better alternative.
The reaction between residual KOH and H2O2 is not detrimental to the results. It only creates oxygen and water, which is of course not harmful. To my understanding there may also be a low concentration of superoxides that form, but they would have a negligible interaction with the plant cells. If anything they would help degrade cell membranes and proteins within the cells.
Heating the mixture helps to further catalyze the oxidation of cell organs, although it does accelerate the natural breakdown of H2O2. This is not of concern as there will be enough H2O2 in the solution to be effective. I don’t have further theoretical details as to why it makes the protocol more effective, but the data suggests that it makes clearer specimens, especially in those with higher tannin concentrations.
I find that it generally helped to do the oxidizing treatment after an initial base treatment, most often after the second treatment. My hypothesis as for why this is is that the strong base “cracks open” most of the cellular components so that the oxidizer can then attack them further. An additional subsequent base treatment clears the remaining proteins. Oxidizer treatments are largely ineffective if they are done as the first step and are moderately less effective if they are done as the final step.
TO BE CONTINUED
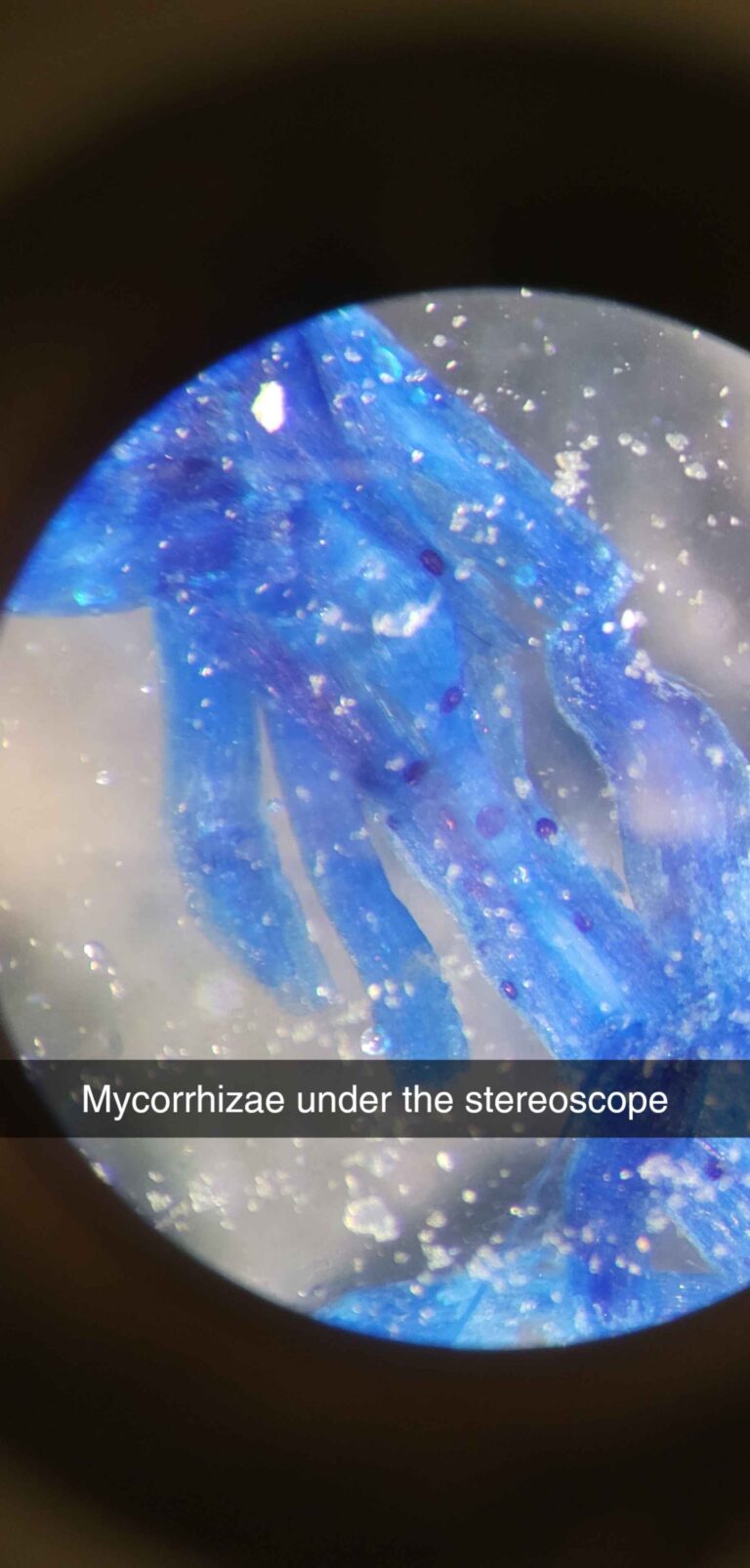
Gallery